|
||||||||||
|
|
||||||||||
|
||||||||||
|
|
||||||||||
|
|
|
|||||||||||||
|
Photon Excitation:In its simplest terms, the PSP method is based on the sensitivity of some luminescent dye to molecular oxygen. After absorbing a photon, a luminescent molecule is placed in an excited state. The molecule then returns to its ground state by emitting a new photon at a longer wavelength, as illustrated below.
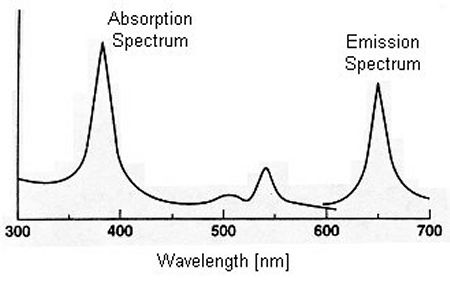
Typical PSP absorption and emission spectra [from McLachlan and Bell, 1995]For some luminescent molecules, however, oxygen quenching allows the molecule to return to a ground state without emitting a photon. Hence, for a given level of excitation, the emitted light intensity from the luminescent molecules varies inversely with the local oxygen partial pressure. Since the mole fraction of oxygen in air is fixed, the oxygen partial pressure is easily converted into the air pressure. Oxygen Quenching:The oxygen quenching process can be mathematically described by the Stern-Volmer relation:
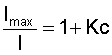
where, for PSP, I is the intensity of the luminescence, Imax is the maximum intensity in the absence of oxygen, K is the Stern-Volmer quenching constant that is characteristic of the luminescent molecule, and c is the concentration of oxygen (O2). Both the Imax and K values are dependent on temperature. Since the luminescent molecules of PSP are suspended in an oxygen permeable binder, Henry's law is needed to relate the oxygen concentration within the binder to that on the PSP surface:
In this equation, S is the Henry's law coefficient (which is dependent on temperature), X is the mole fraction of oxygen in air, and P is the air pressure. By substituting the second relation into the first, the Stern-Volmer relation can be rewritten as:
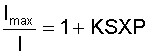
At this point, the relationship between the air pressure and the luminescence intensity is quite clear. As the pressure P increases, the luminescence intensity I must decrease. Unfortunately, this form of the Stern-Volmer equation is not very useful for an experimental setup, since it is usually not practical to obtain the maximum luminescence intensity in the absence of oxygen. A more suitable form of the above equation can be derived by taking the ratio of intensities for two different flow conditions:
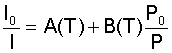
In this equation, the zero subscripts denote a "no-flow" condition, where the pressure is constant over the entire surface. The coefficients A and B are coating sensitivities (which are temperature dependent) that are determined by an experimental calibration. An interesting aspect that arises out of this equation is that the relative intensity is linearly related to the relative pressure. This relationship arises because of the assumption in Henry's law that the concentration of oxygen in the binder is linearly dependent on the pressure over the surface. For most coatings and experimental conditions, the linear assumption is accurate enough. However, for some coatings and conditions, the relationship between the oxygen concentration and the pressure is nonlinear, resulting in a nonlinear relationship between the relative intensities and relative pressures. In these cases, a more general form of Henry's law is needed where S is no longer a function of only the temperature, but pressure as well. When this more general form is used, the coefficients A and B also become functions of both temperature and pressure. As might be expected, this dependency on temperature and pressure is not desirable. Hence, in this situation it is usual to write the coefficient S as a polynomial expansion in pressure, in which the expansion coefficients are only functions of temperature. This manipulation gives a more manageable form of the Stern-Volmer equation:
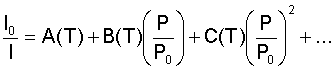
For most cases, the second order approximation shown is adequate enough to describe the behavior of "nonlinear" coatings. Application to Testing:Both of the above relationships are practical equations for use in aerodynamic testing. To use these equations, it is readily obvious that intensity measurements must be taken for flow on and flow off conditions. Since the pressure in the flow off condition is known (P0) and the intensities I and I0 are measured, the pressure P is easily determined from the proper equation. Taking the ratio of the equations that produces these relations has an added benefit besides that of eliminating the need to determine Imax. By taking the ratio of the intensities, the effects of non-uniform illumination and PSP distribution are effectively factored out. This benefit is only valid, however, when the assumption is made that the geometry of the experimental setup and the illumination source remains constant between the measurement of I and I0. Another assumption that needs to be made in the derivation of these equations is that the intensity of the excitation illumination is low enough so that the majority of the luminescent molecules are in their ground state. If the excitation illumination intensity is too high, then a majority of the molecules will be in their excited state, and the coefficients A and B would become functions the illumination intensity as well. A final important note that needs to be made about these equations is that their form will not change if I and I0 are acquired at different temperatures. As long as the flow off temperature is a known constant that is uniform over the entire surface, then its effect on the equation is nothing more than a multiplicative constant that can be incorporated into the coefficients A, B, and C. |


|
Aircraft | Design | Ask Us | Shop | Search |

|
|
| About Us | Contact Us | Copyright © 1997- | |||
|
|
|||